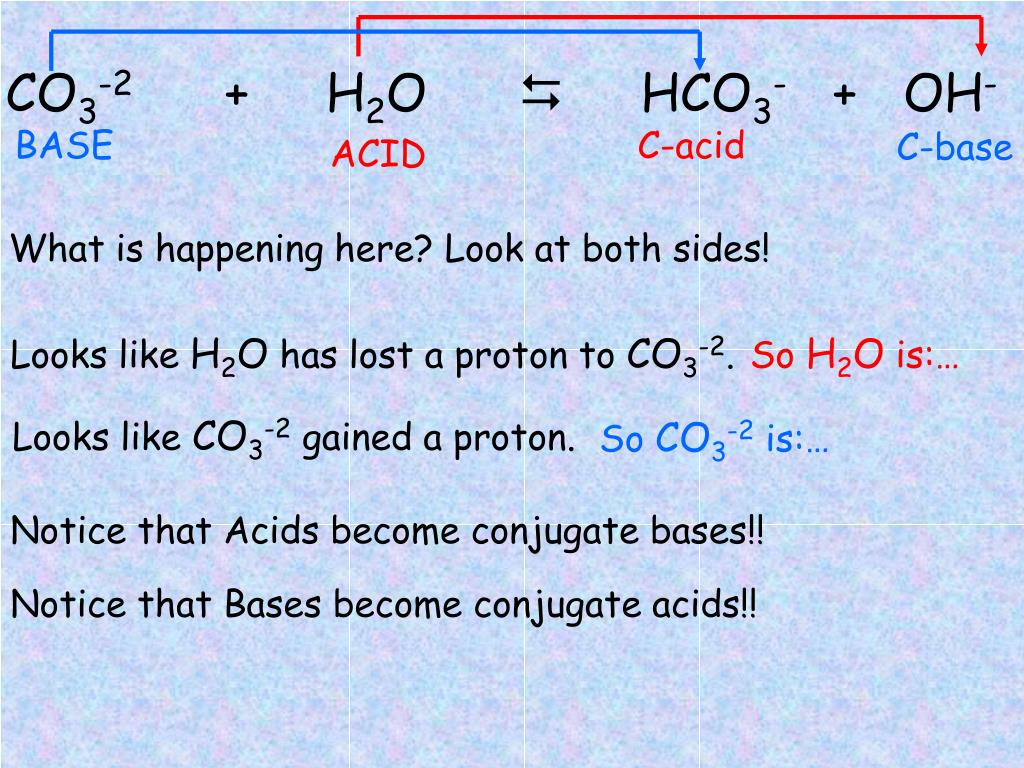
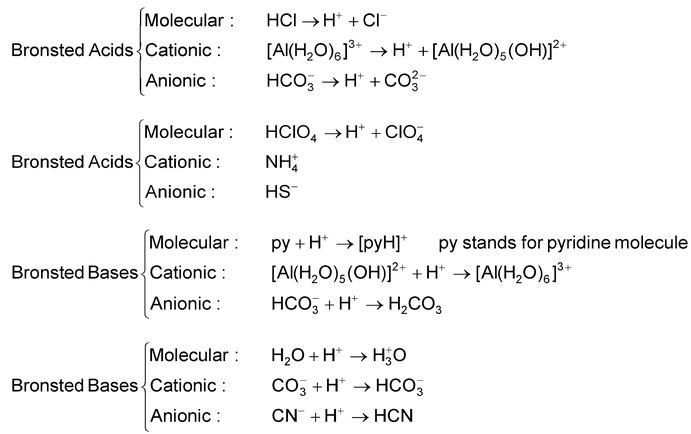
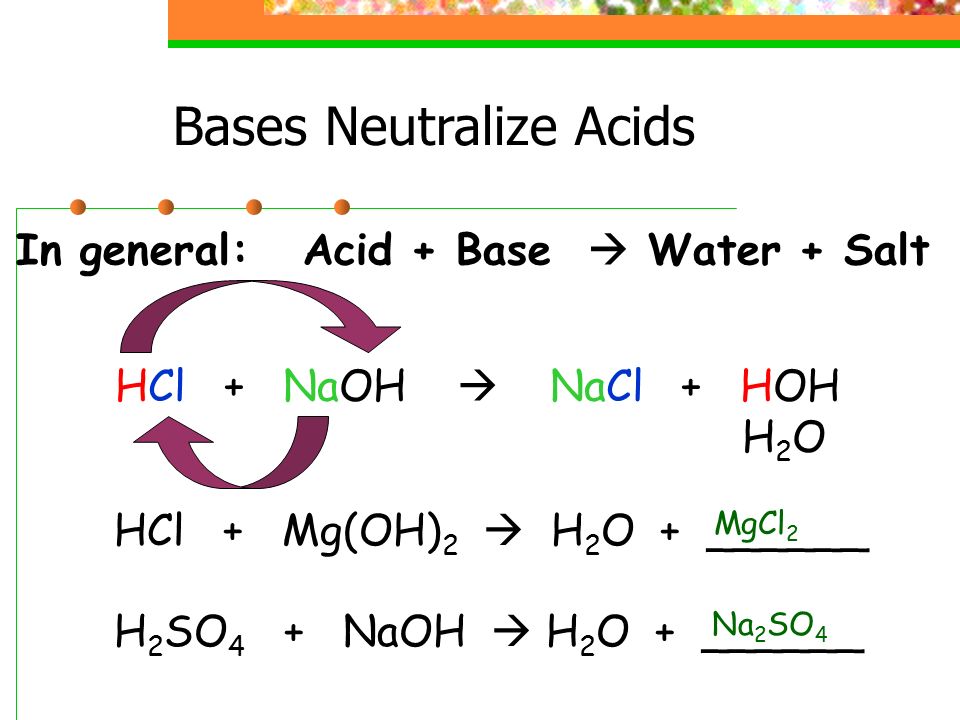
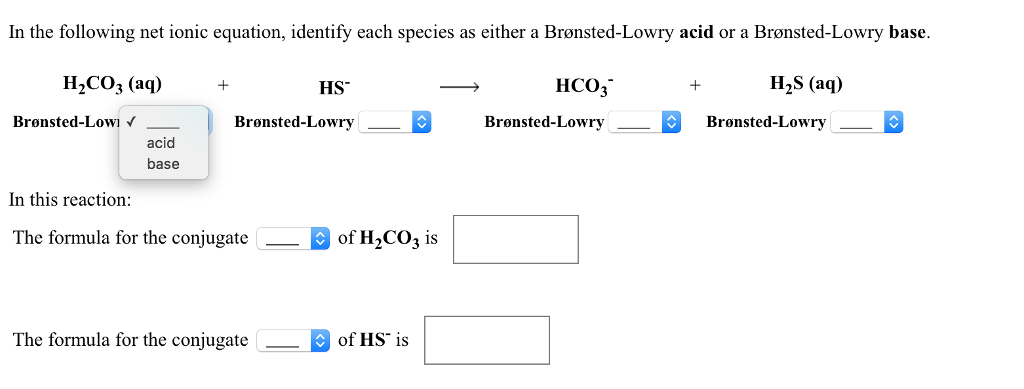
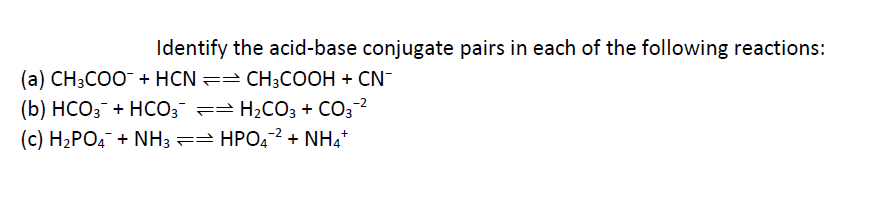SOLVED: Identify the conjugate acid and base pairs in the following acid base reaction: HCO3 NH3 CO3-2 NH4t Acidlconjugate base: HCO3 CO3-2 Baselconjugate acid NH3 NH4 B Acidlconjugate base: CO3-2 / HCO3"

Chemistry Help: The carbonate ion (CO3 2-) acts as a Bronsted base with water and equations - YouTube
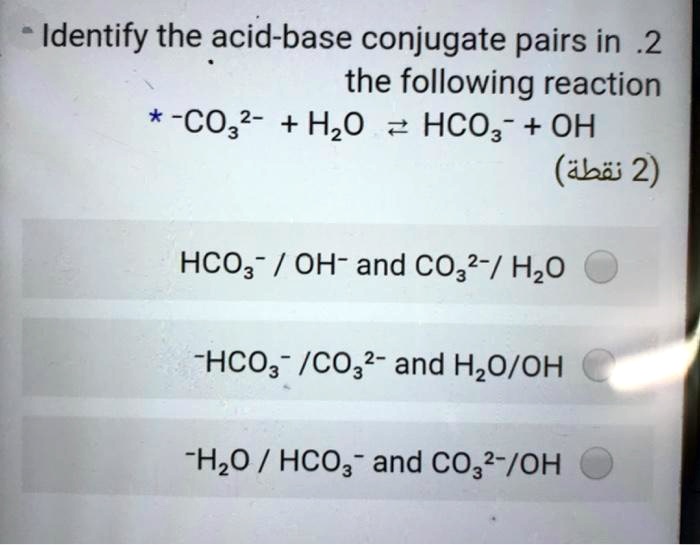
SOLVED: Identify the acid-base conjugate pairs in 2 the following reaction *-CO3 2 - +Hzo 2 HCO; +OH (abii 2) HCO3 1 OH- and CO3?-/ HzO HCO; - /CO32 2 - and HzO/OH Hzo / HCO: and CO3?-/OH