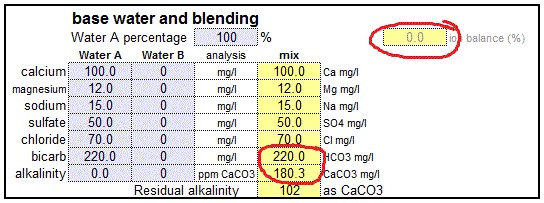
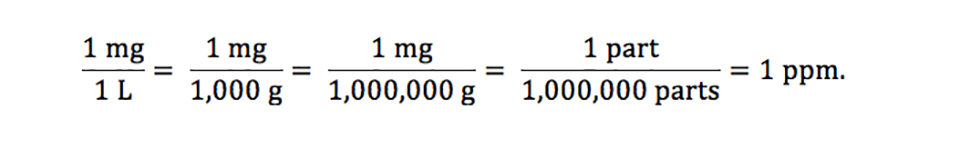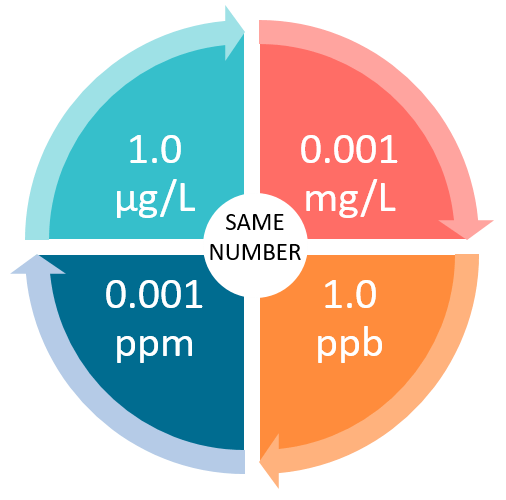Pounds Formula Prepared By Michigan Department of Environmental Quality Operator Training and Certification Unit. - ppt download

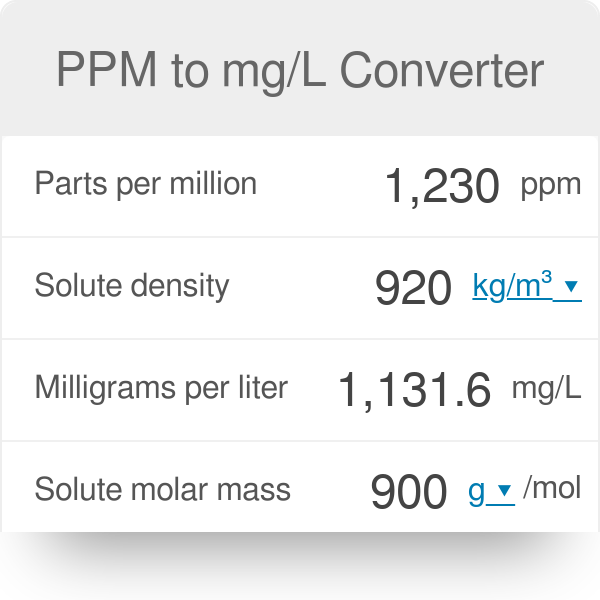
Units Conversion There are 1,000 milligrams (mg) in 1 gram (g) milli = 1 *10-3 milligrams per liter (mg/l) are equal to parts per million (ppm) - ppt download

PPT - mg/L - milligrams per litre (10-3 g per litre) = mg per 1,000,000mg THUS = ppm PowerPoint Presentation - ID:4270418