QCM 2ème partie. N de QUESTIONNAIRE : 006. Examen du 21 Avril Code réservé au CIFMD : - PDF Free Download
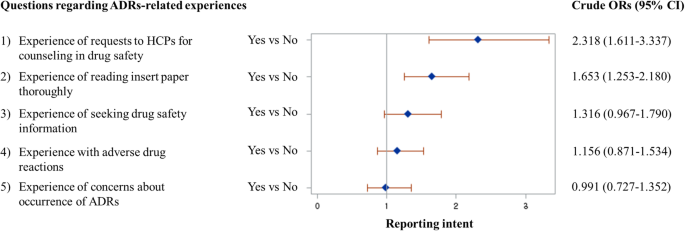
A cross-sectional survey of knowledge, attitude, and willingness to engage in spontaneous reporting of adverse drug reactions by Korean consumers | BMC Public Health | Full Text

PDF) Health professionals' knowledge, attitude and practices towards pharmacovigilance in Nepal | Subish Palaian - Academia.edu

E-pharmacovigilance: development and implementation of a computable knowledge base to identify adverse drug reactions. - Abstract - Europe PMC

PharmActa: Personalized pharmaceutical care eHealth platform for patients and pharmacists - ScienceDirect

PDF) Awareness related to reporting of adverse drug reactions among health caregivers: A cross-sectional questionnaire survey

An evaluation of knowledge, attitude and practice of Indian pharmacists towards adverse drug reaction reporting: A pilot study – topic of research paper in Clinical medicine. Download scholarly article PDF and read

PDF) E-pharmacovigilance: development and implementation of a computable knowledge base to identify adverse drug reactions | Kay Brune - Academia.edu

Are you susceptible to adverse drug reactions? | by Chiao-Feng Lin | DNAnexus Science Frontiers | Medium

Development and validation of a questionnaire for the assessment of the knowledge, management and reporting ADR in paediatrics by healthcare teams (QUESA-P) | BMJ Open
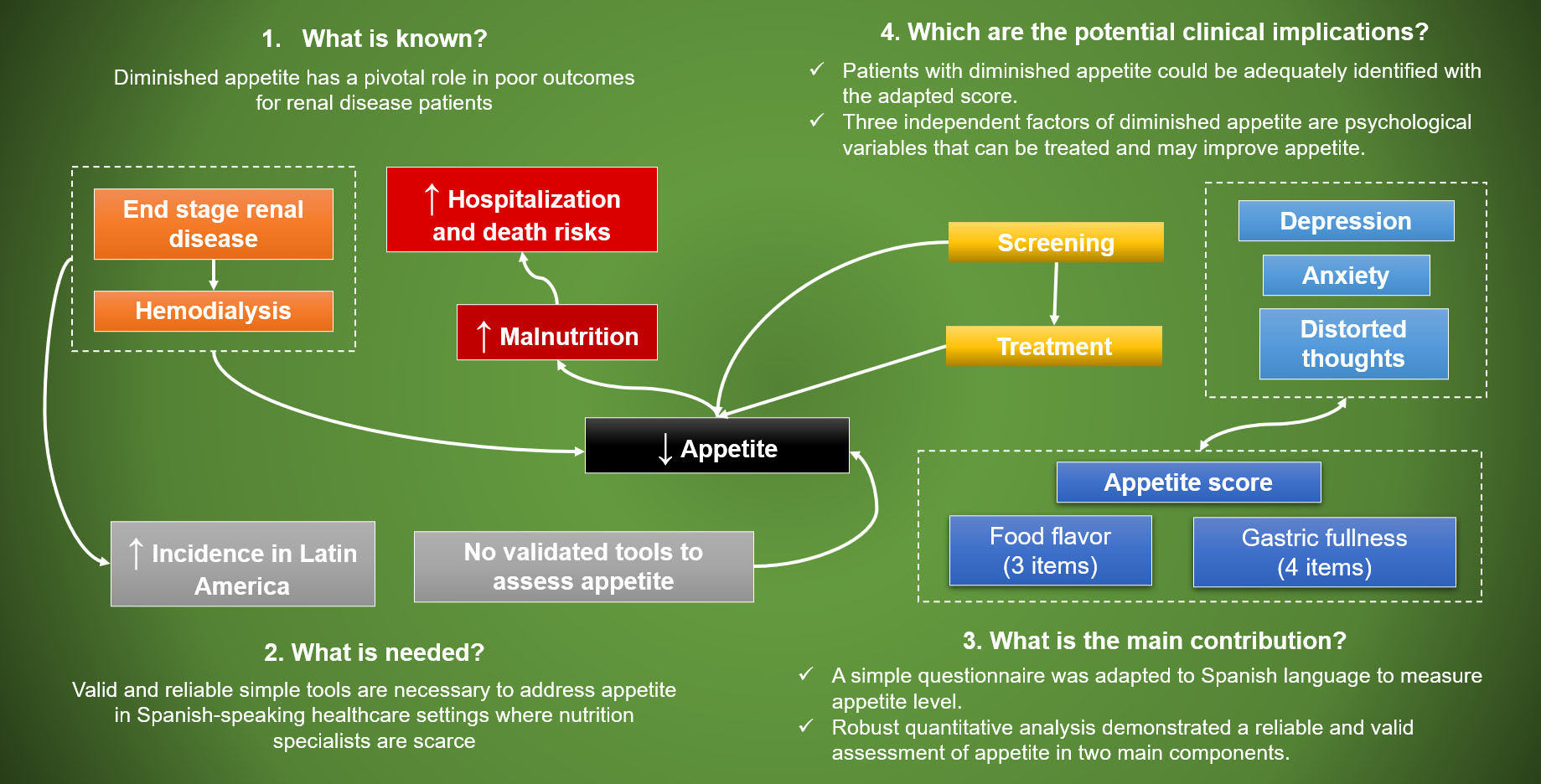
Nutrients | Free Full-Text | Assessment of Factors Related to Diminished Appetite in Hemodialysis Patients with a New Adapted and Validated Questionnaire

Need for an ADR model for project success.pdf - Need for an ADR model for project success 1 de | Course Hero
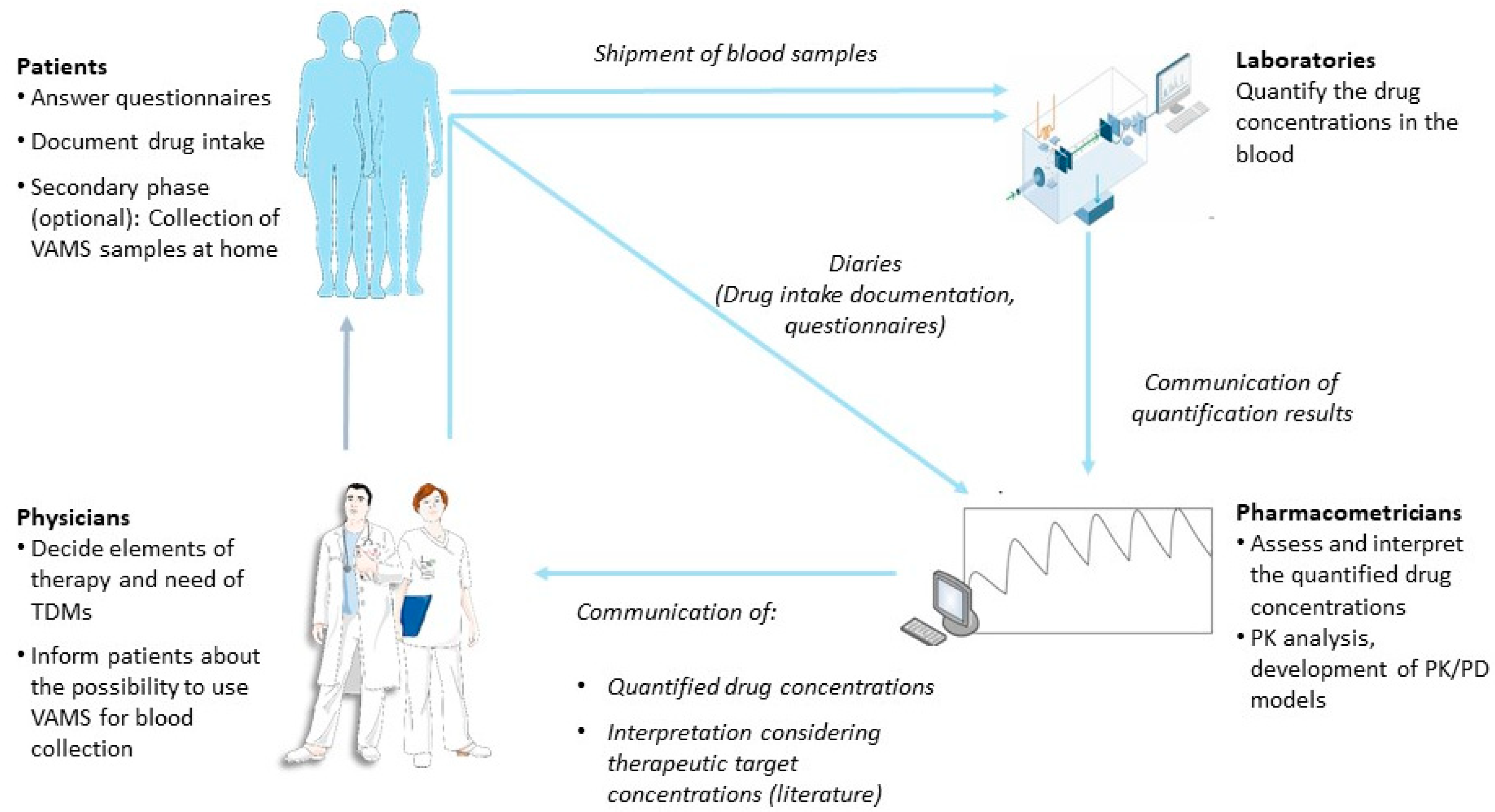
Cancers | Free Full-Text | Developing a Nationwide Infrastructure for Therapeutic Drug Monitoring of Targeted Oral Anticancer Drugs: The ON-TARGET Study Protocol

PDF) Development and Validation of Questionnaire to Assess The Knowledge, Attitude and Practice towards Adverse Drug Reactions Reporting among Healthcare Professionals

Knowledge, attitude and practice among medical students related to Pharmacovigilence | World Journal of Pharmaceutical Sciences - Academia.edu

Advances in Our Understanding of the Interaction of Drugs with T-cells: Implications for the Discovery of Biomarkers in Severe Cutaneous Drug Reactions | Chemical Research in Toxicology



.jpg)